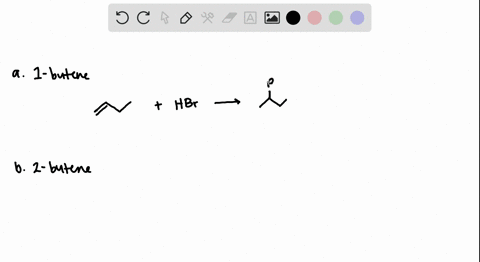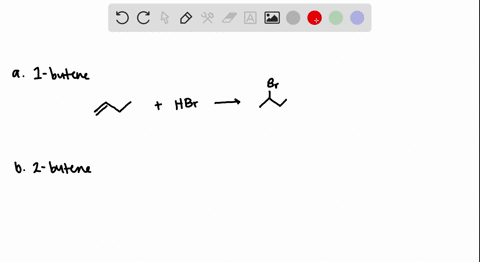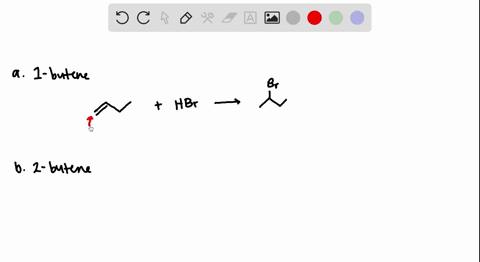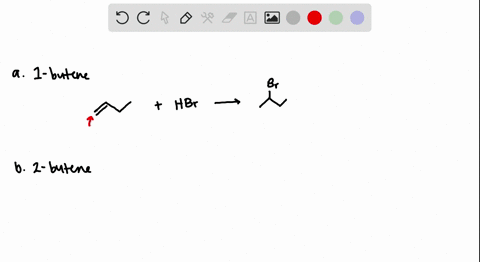
How to predict the product of 1-butene when subjected to an addition reaction with hydrogen bromide (Hbr) - Quora
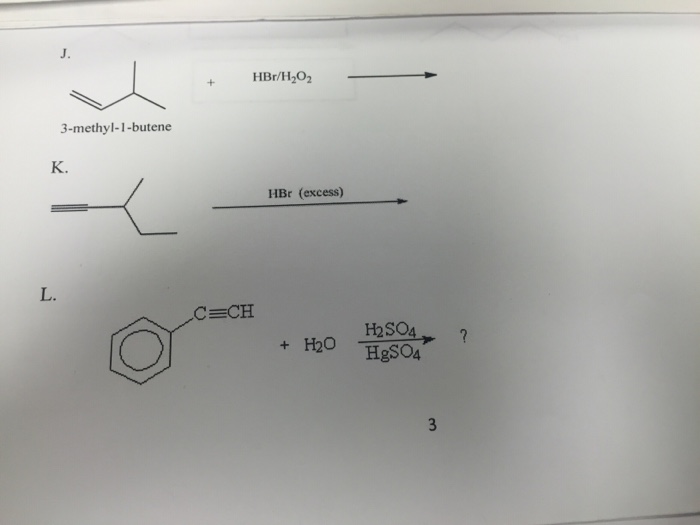
Which one of the following compounds will give in the presence of peroxide a product different from that obtained in the absence of peroxide.A. 1- butene,$HCl$B. 1-butene, $HBr$C. 2-butene, $HCl$D. 2-butene, $HBr$

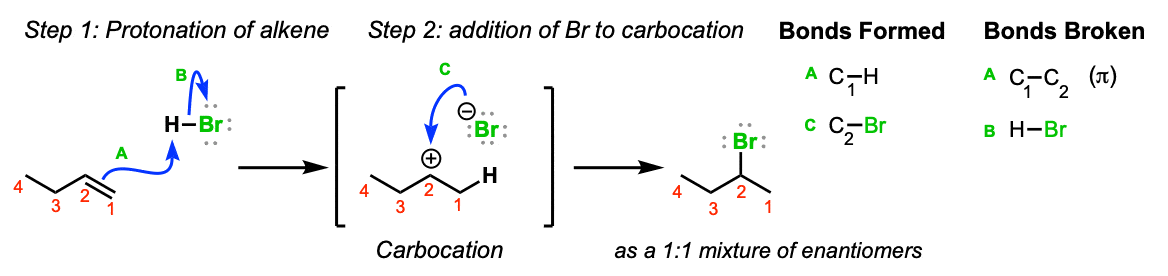
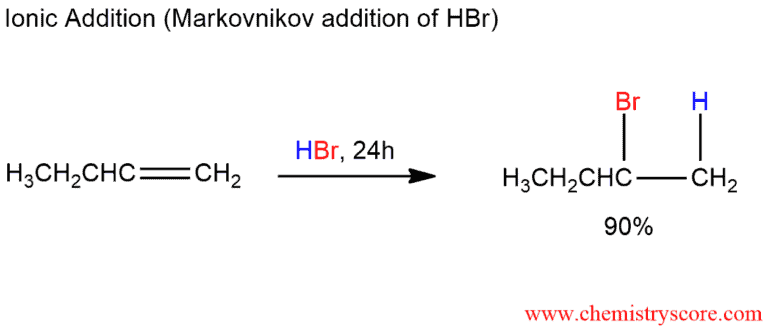
The addition of HBr to 1-butene gives a mixture of products (I),(II) and (III):The mixture consists of :
A:Addition of HBr in buta 1,3 diene gives 3 bromo but 1 ene as major product at low temperature. R::Addition of HBr in buta 1,3 diene gives 1 bromo but 2 ene

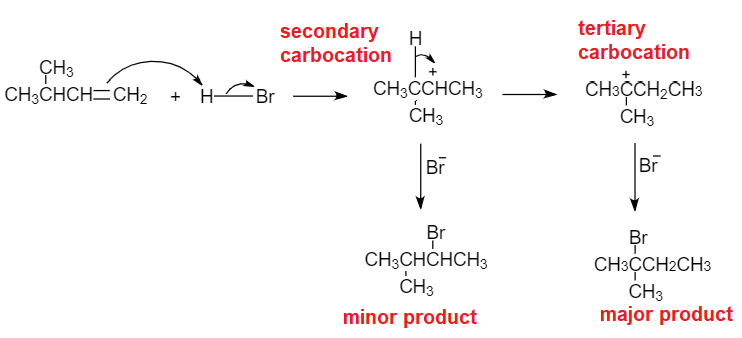
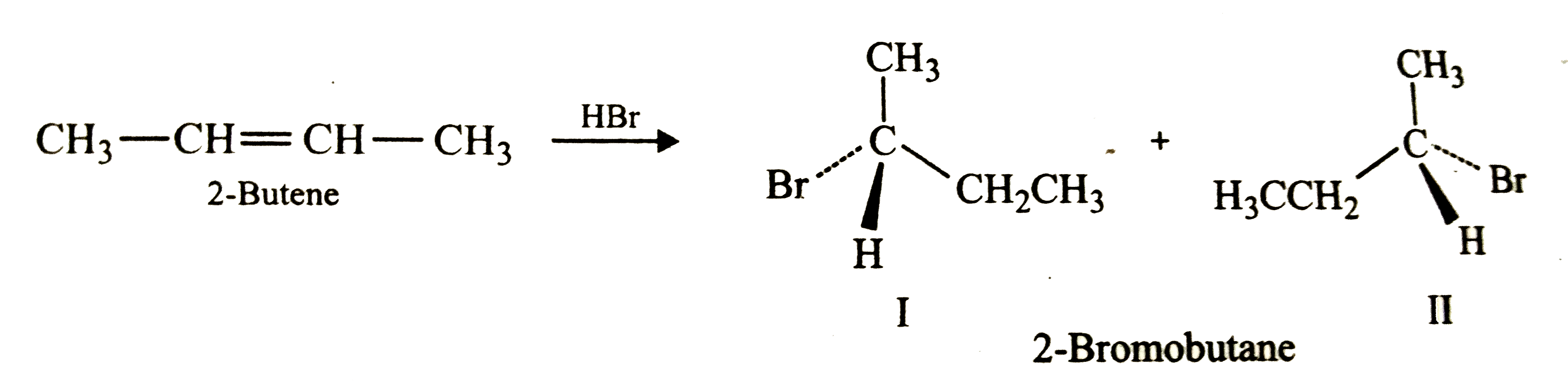
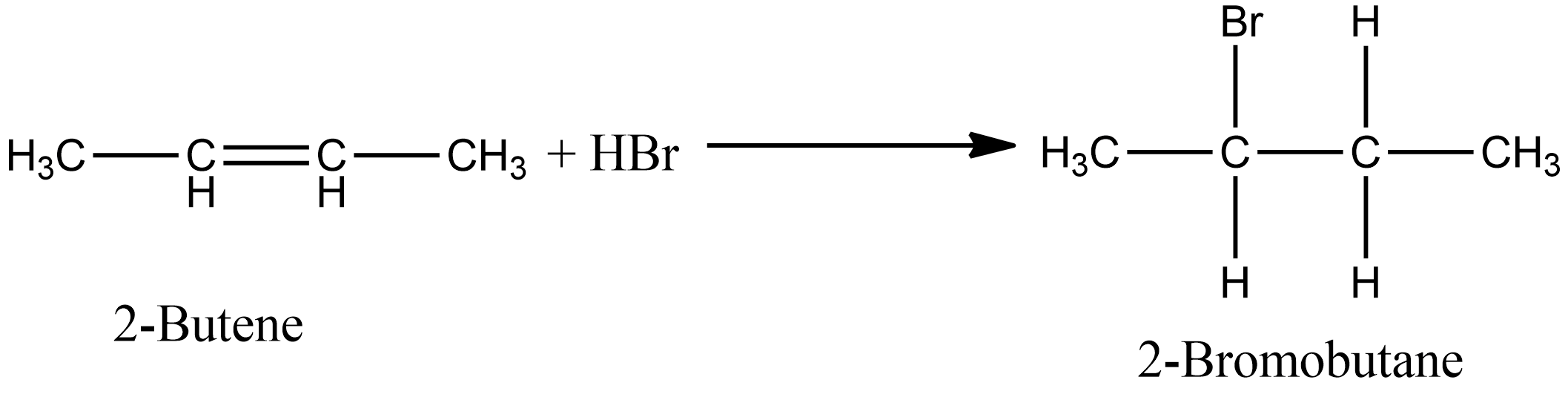
The addition of HBr to 2-butene produces: a. 1-bromobutane b. 2-bromobutane c. 1,2-dibromobutane d. 2,3-dibromobutane e. no reaction | Homework.Study.com

reaction of ALKENES with bromine water but-2-ene but-1-ene cyclohexene hex-1 -ene hex-2-ene ethene propene skeletal formulae ionic electrophilic addition mechanism advanced A level organic chemistry revision notes doc brown
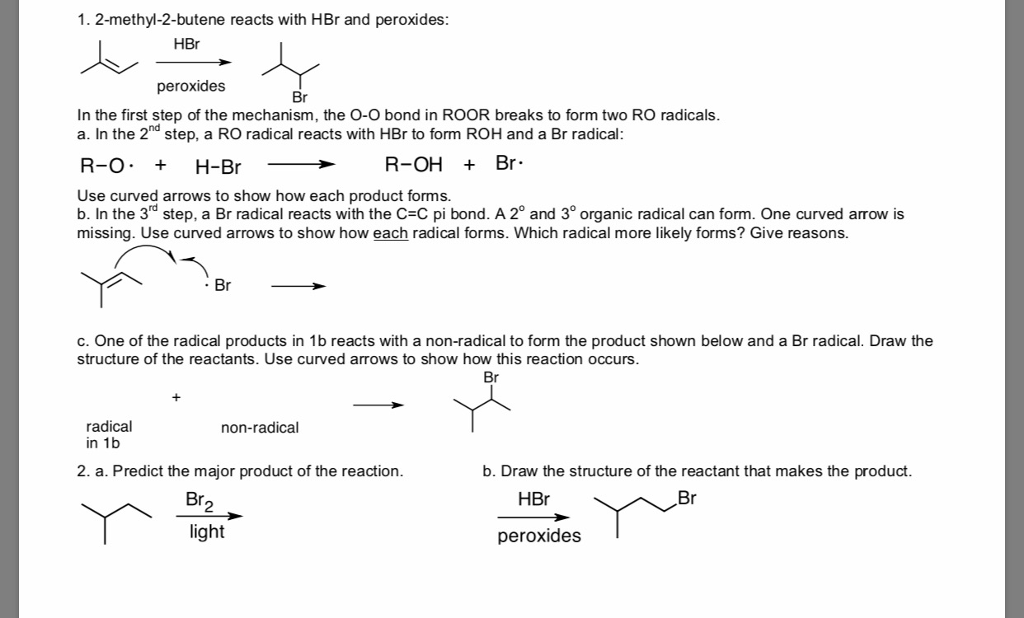
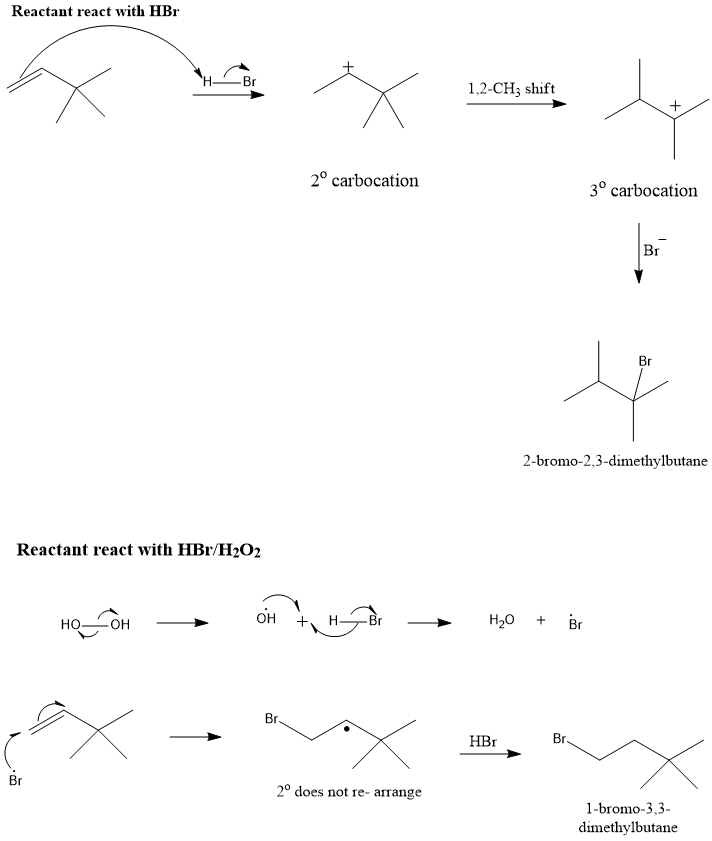
Statement 1: 1-Butene on reaction with HBr in the presence of a peroxide produces 1 -bromo-butane. Statement 2: It involves the free radical mechanism.

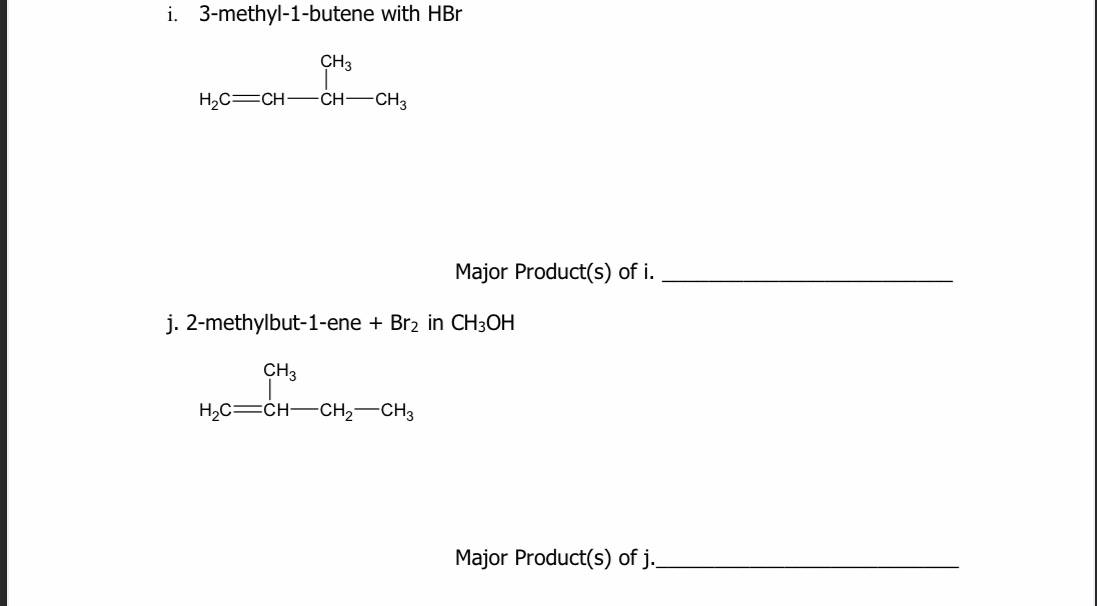
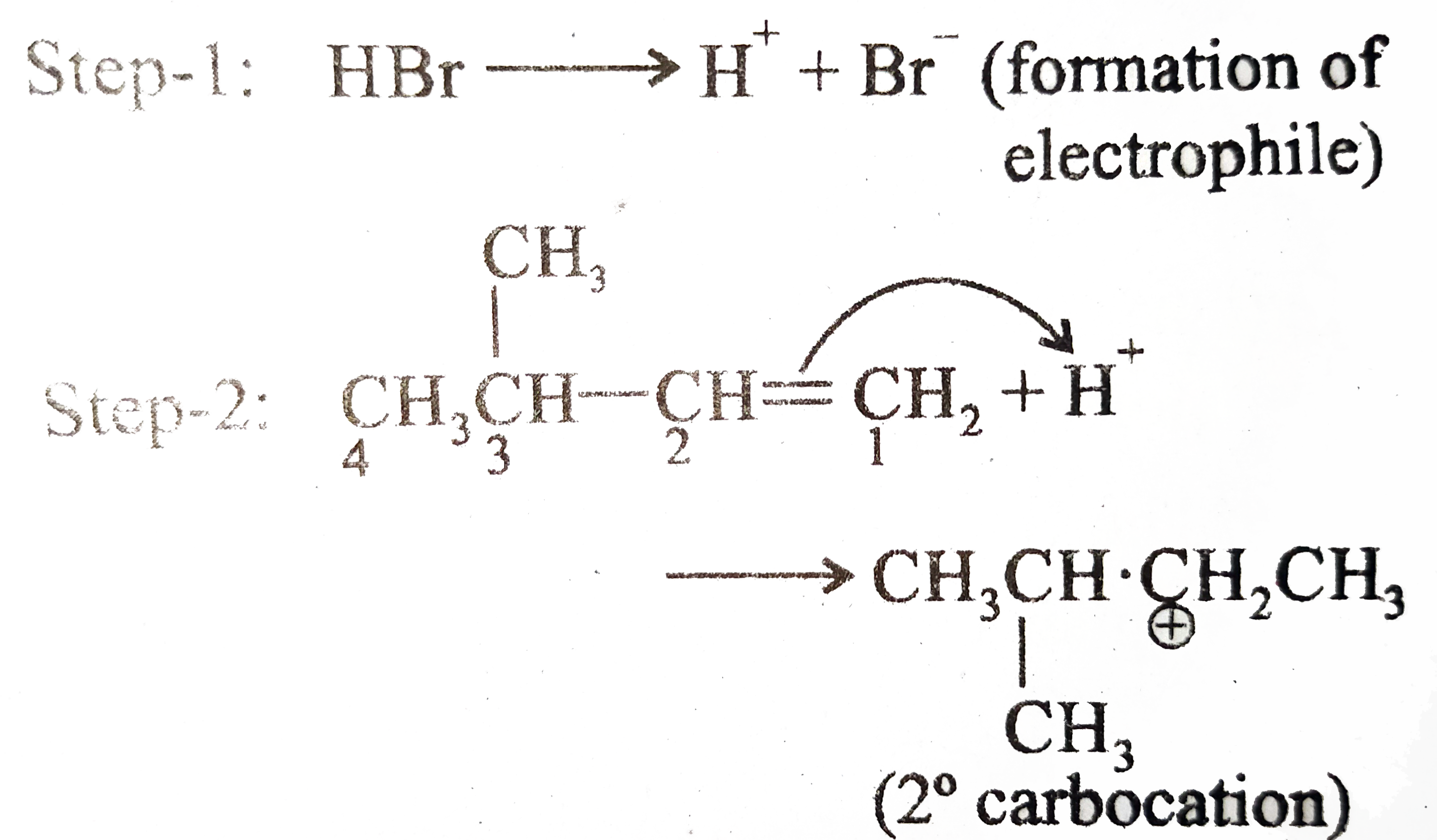
ReasonAddition of HBr on 2-butene follows Markovnikov's rule.AssertionAddition of HBr on 2-butene gives two isomeric products.